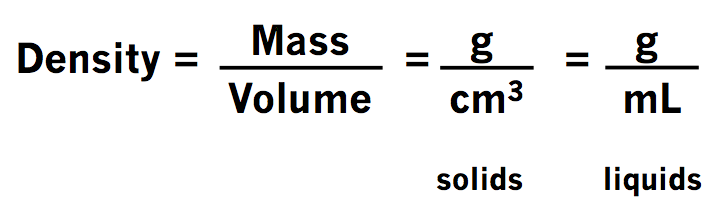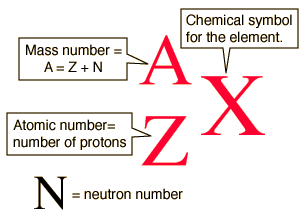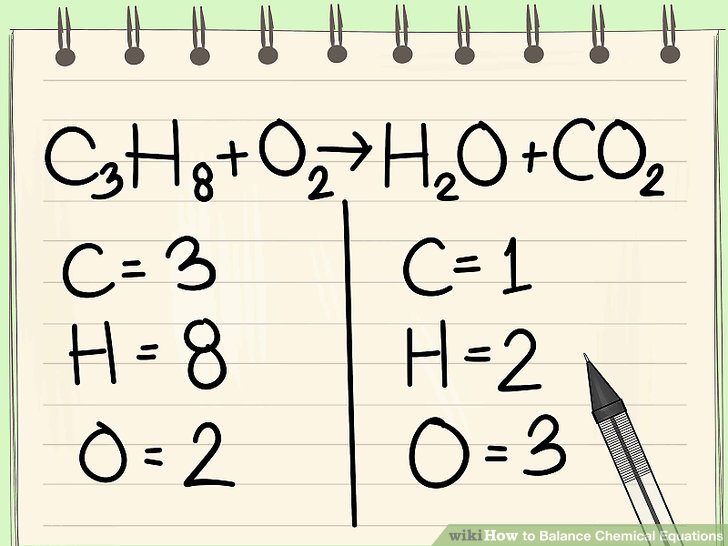Topic 1 - Summer Assignment Part 1
Numbers 1-3 - Please refer to the link below. Significant figures are an important concept throughout this course. Read and practice the rules provided.
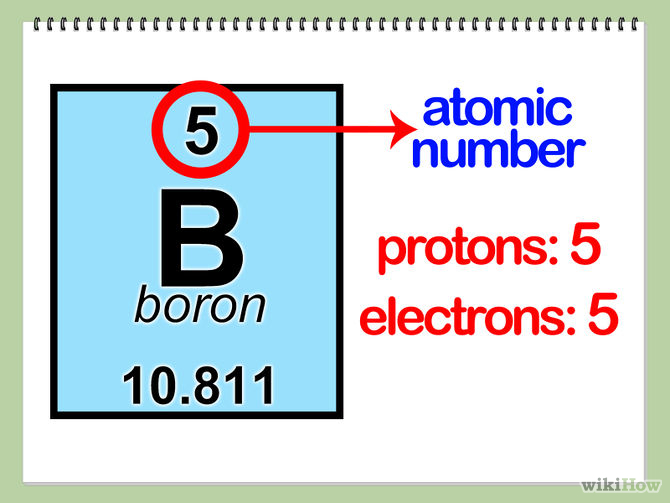
Numbers 4-6 - Please refer to the link below. It will provide you with the set of rules necessary to find common charges and oxidation numbers. The fundamentals of oxidation and reduction will be discussed throughout the year. Provided is also a link to a useful periodic table that you may use throughout the assignment. ALSO: Number six does not have underlined elements. Please find the oxidation numbers for all elements present in each compound.
Numbers 7-8 - Refer to the video below. She does an excellent job reminding you of how to convert between different units (which you may remember from freshman physical science class). The formula for density is provided for number 8.
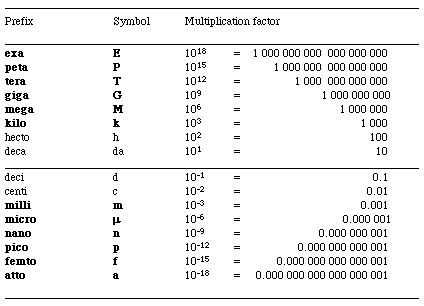
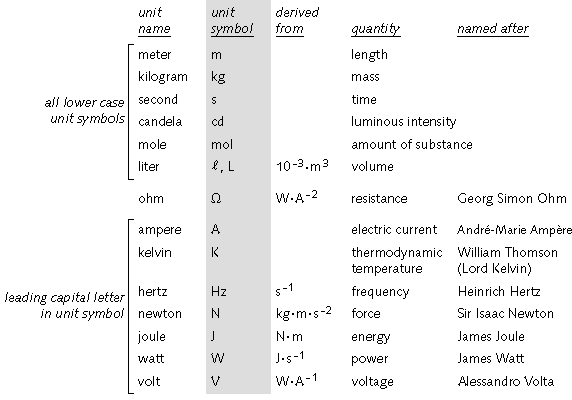
Numbers 9-10 - These questions review units and the metric system. Use the following images as a review.
Numbers 11-12 - Use the following images and links to answer the questions. The PDF file provided will give examples and show the process of number 12 (finding average atomic mass). The periodic table linked earlier will also continue to be useful.
| |||||||
Numbers 13-14 - These problems involve converting between moles and grams. Hopefully, you remember some general ideas about this concept from your freshman physical science. If not, watch the following videos from beginning to end. They may seem a bit long, but they are excellent videos that give you plenty of examples and 2 very clear methods for this process. This process is critical in any chemistry class!
|
|
|
|
Number 15 - Refer to the labeled Periodic Table.
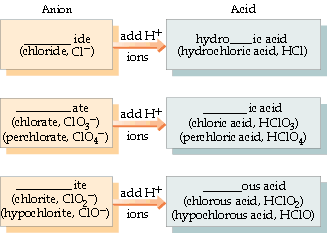
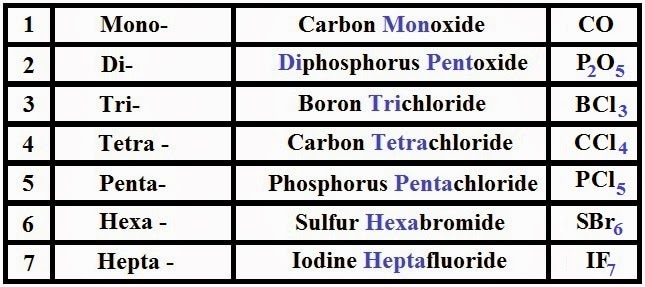
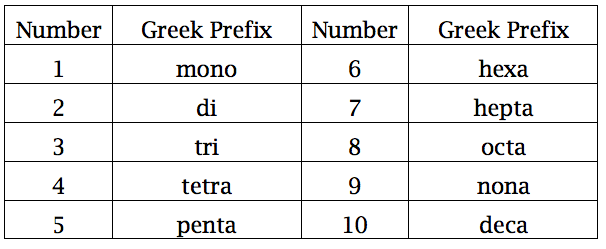
Numbers 16-18 - You probably spent a lot of time with naming compounds your freshman year. The images below outline the rules for acids, ionic and covalent compounds, and hydrates. Included is a list of common ions - please begin to commit them to memory.
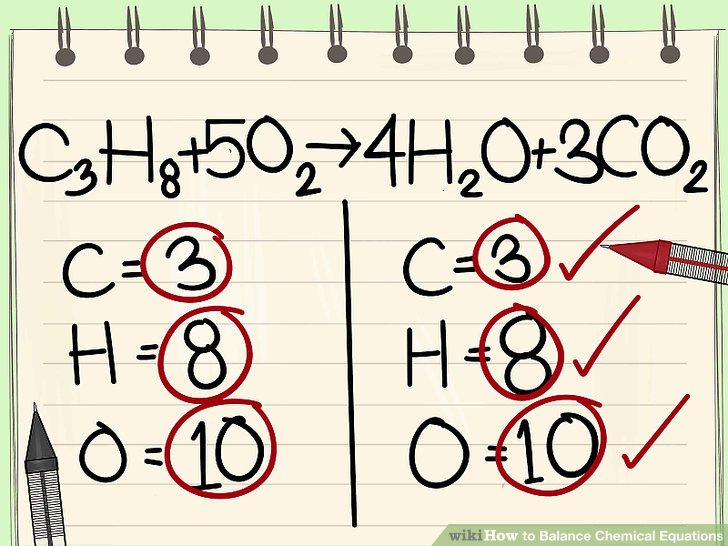
Number 19 - Continue using the naming rules to write out the chemical equations. Remember the Law of Conservation of Matter - you must have the same number of atoms of each element on both sides of your reaction. These must be balanced. Below is an image and a video to help you remember some strategies for balancing equations.
Number 20 - Stoichiometry (using mole ratios) is another critical concept in chemistry. The following video does a great job of introducing you to the idea. You did quite a few of these your freshman year. Watch the video first, then attempt the problems.
Numbers 21-24 - These are challenging problems that apply what you've already done throughout this same packet. I would like you to give these a try using all the resources on this page and any of your prior work in this packet. You can do it!
Numbers 25-27 - These problems further use molar masses and ratios. The link below gives a specific example on how to go about these problems.
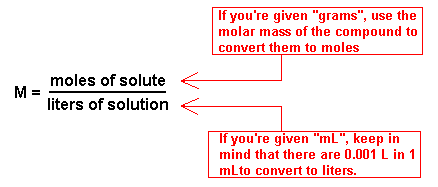
Numbers 28-30 - Molarity begins to lead to solutions chemistry and is one of the most applied ideas in chemistry lab. Below, the formulas for molarity and dilution (number 30 is a dilution problem) are provided. Keep in mind you will need to have all units in moles and liters, NOT grams or mL.